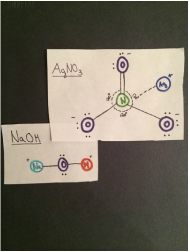
 AgNO3 interacts with NaOH
AgNO3 interacts with NaOH
- The intermolecular forces acting on AgNO3 and NaOH are dipole-dipole interaction, hydrogen bonding, and dispersion forces. Both the NaOH and AgNO3 arr polar molecules so it is dipole-dipole. The positive hydrogen on the NaOH attracts to the negative oxygen on AgNO3 so there is hydrogen bonding. Dispersion forces act on any two adjacent molecules in a liquid.
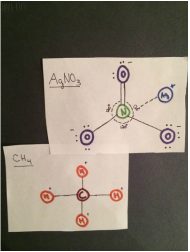 AgNO3 interacts with CH4
AgNO3 interacts with CH4
- The intermolecular forces acting on AgNO3 and CH4 are hydrogen bonding and dispersion forces. CH4 is non- polar so dipole-dipole interaction can not occur. The positive hydrogen on CH4 attracts to the negative oxygen on AgNO3 so there is hydrogen bonding. Dispersion forces act on any two adjacent molecules in a liquid.
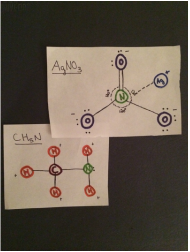 AgNO3 interacts with CH5N
AgNO3 interacts with CH5N
- The intermolecular forces acting on AgNO3 and CH4 are dipole-dipole interaction, hydrogen bonding, and dispersion forces. CH5N and AgNO3 are both polar molecules so dipole-dipole interaction occurs. The positive hydrogen on CH5N attracts to the negative oxygen on AgNO3 so there is hydrogen bonding. Dispersion forces act on any two adjacent molecules in a liquid.
